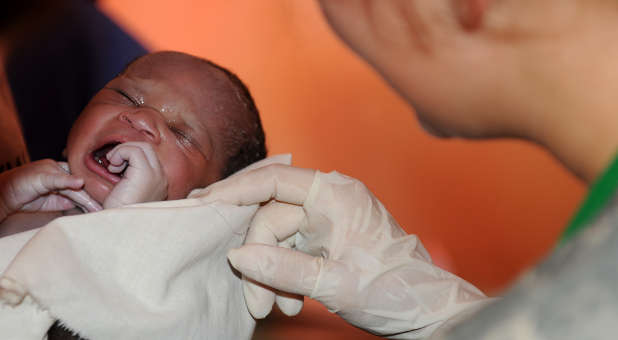
September is Newborn Screening Awareness Month, which educates parents about tests that help diagnose diseases and disorders early in their baby’s life.
But during this awareness month, Citizens’ Council for Health Freedom wants to inform Americans about how the federal “common rule” actually strips parents and the nation’s youngest citizens of their rights.
President Donald Trump has wisely put a hold on the final common rule (“Federal Policy for the Protection of Human Subjects”), which was published by the Obama administration on Jan. 19, 2017. But what will happen in January 2018, when the rule is supposed to go into effect?
CCHF’s concerns about the common rule in its present state include that parental consent requirements for research using newborn DNA, which became federal law in 2014, and the law’s “human subject” designation for newborn DNA are not replicated in and protected by the common rule.
“This 2014 federal law called a child’s newborn bloodspot a ‘human subject’ and prohibited all waivers to the parental consent requirement,” said CCHF president and co-founder Twila Brase. “But the law dictates that this protective language would disappear from federal statute after the final common rule was published because, as CCHF was told, National Institutes of Health leadership had committed to include the 2014 statutory language in the final common rule.”
Brase added that CCHF is pleased the Trump administration has put the common rule on hold, but wonders what will happen to parental rights and patient privacy once Jan. 19, 2018, rolls around—the date the rule was originally supposed to take effect.
“To underscore the importance of this hold, CCHF has advised the president that the genetic privacy of the four million children born each year is at stake,” Brase said. “Under the final common rule, the DNA of newborn citizens could be stored, used, accessed, analyzed and distributed by states without parent consent. These children will grow into voting adults who may or may not learn their genetic privacy rights were lost at birth. As an example of some of the intrusions over the years, newborn DNA has been used for barter in Texas, shared with the CDC for research in Minnesota and sent to the U.S. military to create a national mitochondrial DNA registry.”
In December 2014, President Barack Obama signed the Newborn Screening Saves Lives Reauthorization Act (NSSLRA), which included language CCHF worked with the office of Sen. Rand Paul and the Senate Steering Committee to develop. Hospitals routinely prick the heels of newborns and send the dried blood spots to state public health departments for government newborn genetic screening programs. The language in the NSSLRA required parental consent for the use of these blood spots for federally funded research. Unbeknownst to most parents, many states store the blood spots in perpetuity without parental consent.
When the rule was finalized, however, it did not include parental consent requirements or any other part of the protective language of Sen. Paul’s NSSLRA amendment. In fact, it specifies that the 2014 NSSLRA language “will no longer apply after the effective date of this rule, Jan. 19, 2018.” And, specifically, the definition of “research” excludes “public health surveillance activities, including the collection and testing of information or biospecimens conducted, supported, requested, ordered, required or authorized by a public health authority.”
CCHF first discovered the unconsented storage, use and sharing of newborn DNA by state health departments in Minnesota in 2003. Storage began in 1997, solely as an executive decision. No law authorized it. CCHF’s efforts against “baby DNA” warehousing have led to three successful parent lawsuits in Texas and Minnesota, changes in laws and rules, media coverage by Science and Nature and a television report disclosing 666 banker boxes of stored newborn dried blood spots in Indiana (23 years of newborns), now available to researchers without consent.
“But most Americans still have no idea that states are storing, using and sharing newborn DNA and conducting genetic research on children,” Brase says. “These children who later become adults are also unaware. This acquisition of private property happens when new parents are exhausted, exhilarated, in pain and according to newborn screening research, in a ‘fog.’ They think every lab test is a hospital test. They don’t know their baby’s DNA is being sent to the government.
“Most Americans also do not realize that the federal government in 2013 provided $25 million to four institutions for five-year research projects to sequence the genomes of newborn children and study the legal, ethical and social ramifications of genetic detailing at birth,” Brase added.
The Trump administration, says CCHF, now has an opportunity to right this wrong at the federal level by revising the common rule to include all the protective parental consent and “human subjects” language of the Newborn Screening Saves Lives Reauthorization Act of 2014. CCHF also recommends that Health and Human Services funding for state newborn genetic screening programs be contingent on the state’s annual attestation that no newborn dried blood spots have been stored or analyzed (beyond the child’s newborn screening test) or used, shared or sequenced without the consent of the child’s parents. Consent acknowledges that each individual’s genetic code and genetic traits are private and under the parent’s and, eventually, the adult child’s purview.
Read CCHF’s letter to Trump and Secretary Tom Price on the Common Rule here.
For more information about CCHF, visit cchfreedom.org, its Facebook page or its Twitter feed @CCHFreedom. Also view the media page for CCHF here. {eoa}
See an error in this article?
To contact us or to submit an article